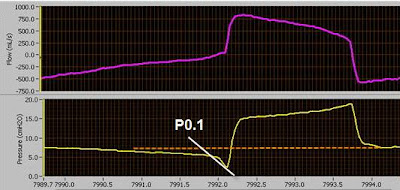
- Click on the picture for a larger image.
Notice in the above pictured graph the dramatic increase in both p0.1 (blue) and work of breathing (yellow) after switching the patient to a low tidal volume strategy.
Maintaining this strategy in this referenced patient would put them at risk for:
- muscle fatigue
- ventilator asynchronies
- higher sedation use
- prolonged mechanical ventilation.
note-
"Forcing a tidal volume of 6 ml/kg on every patient can result in marked cardiovascular instability, as a result of the patient fighting the ventilator for a larger tidal volume. This increases the patients effort, discomfort, oxygen consumption, carbon dioxide production, and increases the complexity of managing the patient" (Kacmarek, R 2007).
If the p0.1 is too high
- Decrease the trigger sensitivity.
- Shorten rise time (faster).
- Increase Flow rate.
- Change to a pressure mode.
- Increase the Driving pressure.
- Consider Proportional Assist Ventilation or Adaptive Support Ventilation.
Reference
Grooms, D. (2006). P0.1 Trending to Evaluate Target Vt Settings. Excel Graph. Norfolk General Hospital. Norfolk, VA.
Steinberg, K. & Kacmarek, R. (2007). Should Tidal Volume be 6 ml/kg Predicted Body Weight in Virtually All Patients with Acute Respiratory Failure? Respiratory Care. 52 (5): 556